The (Revised) Fibromyalgia Impact Questionnaire
The Revised Fibromyalgia Impact Questionnaire assesses how this condition affects daily life and symptoms. Clinicians use this tool to track patient progress and guide treatment decisions.


(Revised) Fibromyalgia Impact Questionnaire
The Revised Fibromyalgia Impact Questionnaire (FIQR) assesses health status in patients with fibromyalgia. It evaluates physical function, symptom severity, and overall impact on daily life. Updated for easier scoring, this tool helps clinicians track pain, fatigue, and stiffness levels accurately.
Category
Disease
Source
What is (Revised) Fibromyalgia Impact Questionnaire

The Revised Fibromyalgia Impact Questionnaire (FIQR) is a top tool for measuring how fibromyalgia affects daily life and overall health status. It updates the older FIQ to capture patient experiences better. The tool splits into three distinct domains covering physical function, overall impact, and symptom severity. Patients rate items on a simple zero to ten numeric scale, making scoring much easier to handle. It looks at issues like pain, energy, stiffness, and sleep quality, plus how hard it is to do routine tasks like shopping or cleaning the house. Clinicians like that it takes under two minutes to finish, so it fits well into busy practices. It spots small changes in a patient's condition, making it great for tracking progress over time. This helps doctors tweak care plans to get better results for their patients while keeping things practical.

The Revised Fibromyalgia Impact Questionnaire can be scored using one main approach based on weighted domains. It looks at three areas: function, overall impact, and symptoms. You score the twenty one items from zero to ten. To calculate, sum the nine function items and divide by three. Add the two overall impact items just as they are. Then, sum the ten symptom items and divide by two. Combine these weighted scores for a total between zero and one hundred. Higher numbers mean the patient is doing it tough with their condition.

Advantages
Measures healthcare intervention results systematically.
Strengthens evidence through systematic measurement approaches.
Develops personalised strategies based on assessment data.
Simple to understand and complete for patients.
Related Instruments
Screens for neuropathic pain components.
Predicts long-term disability risk in musculoskeletal pain.
Assesses symptoms and function in lumbar spinal stenosis.
Streamline (Revised) Fibromyalgia Impact Questionnaire assessments with WeGuide
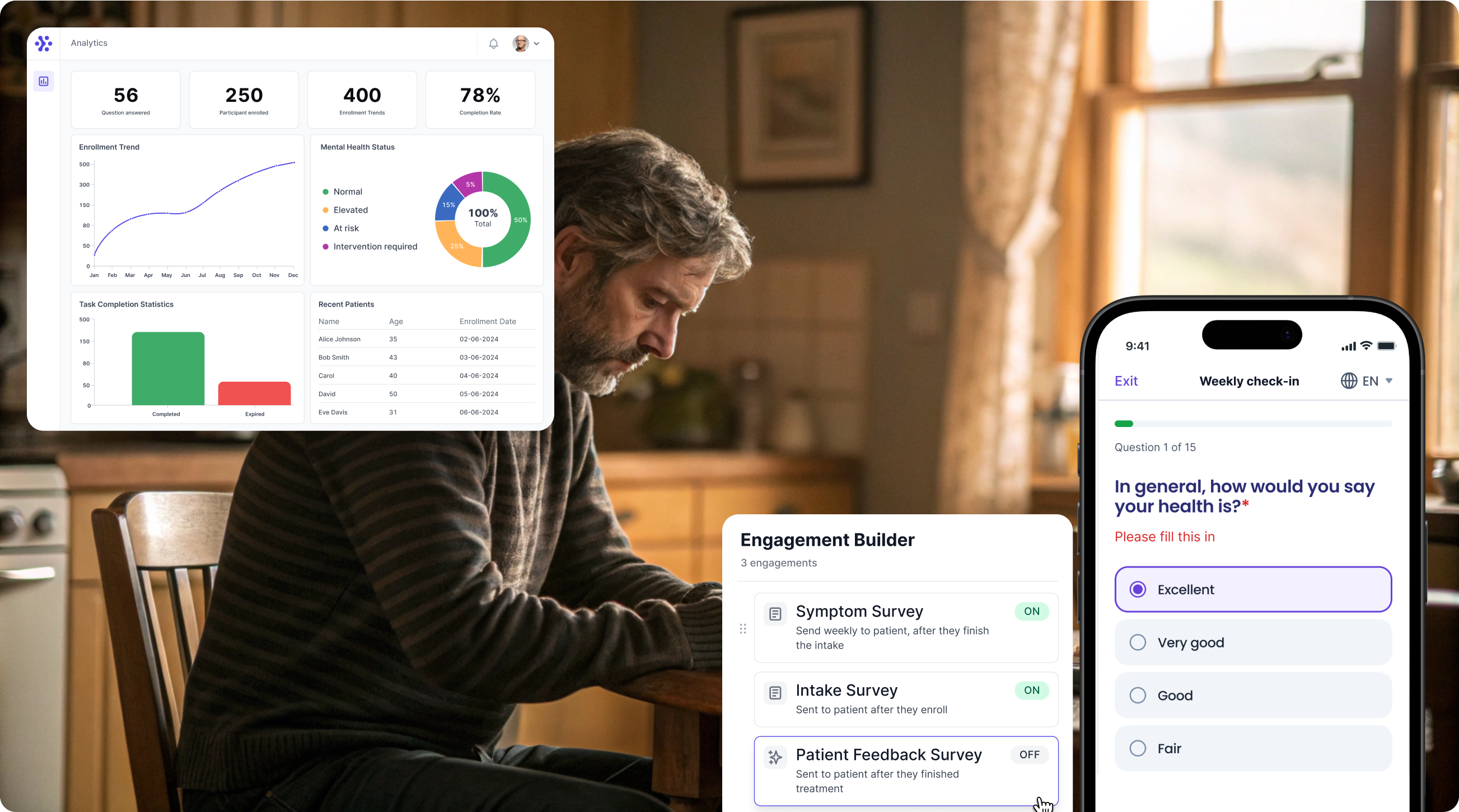

Support Your patient in no time - all under your own brand
Our platform combines the knowledge of 100+ digital health solutions built, letting you easily build your patient engagement app for research or clinical use. Collect PROMs, support patients on waiting lists, or gather vital signs data. This is how:
Learn MoreProgram Builder
.png)
Whitelabelling
.svg)
.svg)
Turn on components, dependent on your usecase
.svg)
Support The Full Journey
.png)
.png)
%20(1).png)
.png)
It’s your turn
Every WeGuide project, saves researchers and clinicians - 9 months and $450,000 - creating impactful digital health solutions.




