eConsent Software for Clinical Trials
Manage electronic informed consent effortlessly with WeGuide. Design interactive consent forms, track participant status in real time, maintain a complete audit trail, and handle protocol amendments with ease. Whether your study is site-based, remote, or hybrid, WeGuide keeps your consent process compliant and participant-friendly.
By clicking Organise a demo, you're confirming that we can contact you to set up a demonstration.

Used by leading research and medical institutes.








%201.avif)










What is eConsent?
eConsent (electronic consent) is the digital process of obtaining informed consent from participants in clinical trials and health research studies. Instead of relying on traditional paper based informed consent forms (ICFs), eConsent uses electronic media, including interactive text, videos, audio, images, and comprehension quizzes to convey study information and capture a participant's agreement through an electronic signature.
The goal of eConsent in clinical trials is to improve participant comprehension, reduce protocol deviations related to incomplete or incorrect consent documentation, and create a verifiable audit trail that satisfies regulatory requirements. eConsent can be administered on-site at a clinical research facility, remotely from a participant's home, or in hybrid settings, making it a core component of both traditional and decentralized clinical trials (DCTs).
Unlike paper consent, electronic informed consent software provides real-time version control, automated reconsenting workflows when protocols change, and digital tracking that eliminates the data ambiguities commonly associated with paper forms.
%20(1).png)
How WeGuide eConsent Works
Build Your Informed Consent Forms
Create engaging eConsent forms using text, video, audio, and comprehension quizzes. Add an electronic signature step for final approval. WeGuide's form builder requires no coding design your ICF exactly the way your IRB or ethics committee has approved it.




Integrate Consent Into Your Study Program
Use WeGuide's program builder to add eConsent forms at the appropriate stage of the participant journey. Define when consent is required at screening, enrollment, or before specific study procedures and the platform handles the rest.




Participants Review and Sign
Participants access the eConsent form on any device. They review interactive materials at their own pace, complete comprehension checks, and provide their digital signature. Support for Legally Authorized Representatives (LARs) and multiple signatories ensures compliance with all participant populations, including paediatric and cognitively impaired groups.




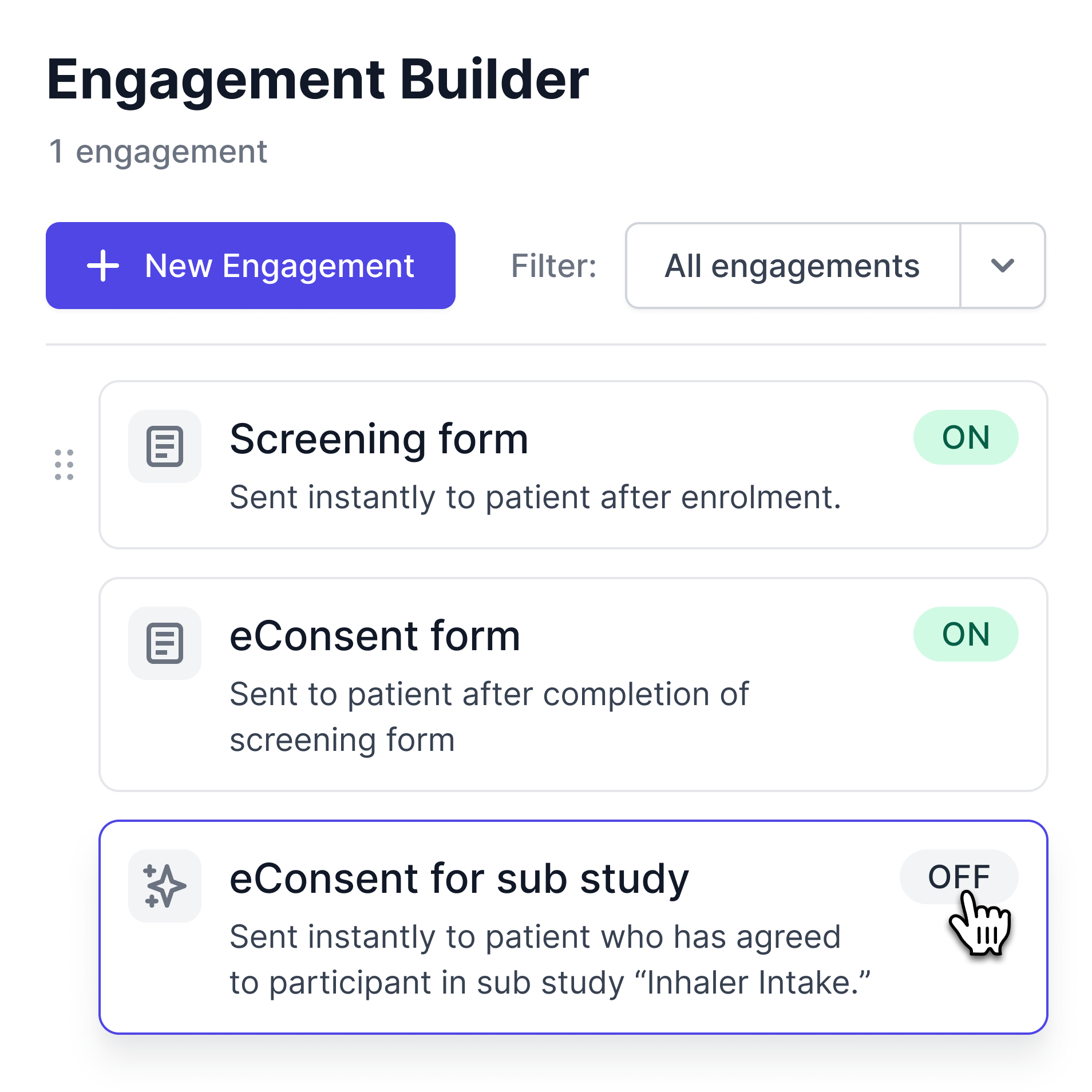
Dynamically Update Your Consent Flow
When protocols change or sub-studies are launched, add new consent steps without disrupting your existing workflow. WeGuide automatically notifies affected participants and tracks re-consent status in real time, ensuring your study stays compliant with ICH-GCP and local regulatory requirements.




Ongoing Patient Engagement
Keep participants informed and engaged beyond consent. Use WeGuide's built-in tools for news updates, educational content, symptom tracking, and remote monitoring — all within the same platform that manages their consent.




Patient engagement
Build and maintain a patient community with News, Education, and Symptom Tracking.











Built for Compliance
WeGuide's eConsent software is designed to meet the regulatory standards required for clinical trials and health research across the globe. Our platform supports compliance with
21 CFR Part 11:
FDA requirements for electronic records and electronic signatures
ICH-GCP (E6 R2:
International guidelines for good clinical practice in clinical research
HIPPA:
Protection of participant health information in US-based studies
TGA (Class 1):
Australian Therapeutic Goods Administration classification for clinical software
ISO 27001:
International standard for information security management
GDPR:
European data protection requirements for studies involving EU participants
Every eConsent interaction generates an immutable, time stamped audit trail including form access timestamps, quiz responses, signature events, and version acknowledgements. This provides sponsors, CROs, and regulatory auditors with the documentation they need to verify that informed consent was obtained correctly and completely.
.png)
Questions
Find answers to frequently asked questions about eConsent for clinical trials, including how electronic informed consent works, compliance requirements, and how to get started with WeGuide.
Contact usWhat is eConsent, and how does it work?
eConsent (electronic consent) is the digital process of obtaining informed consent from participants in clinical trials. With WeGuide, participants review interactive consent forms — including text, video, audio, and comprehension quizzes — and provide their electronic signature digitally. The entire process creates a verifiable audit trail, ensuring participants fully understand the study before enrolling.
Can I customize my eConsent forms?
Yes. WeGuide's form builder lets you customize every aspect of the consent process. Add video walkthroughs, audio explanations, comprehension quizzes, and conditional logic to create an interactive informed consent experience tailored to your study's specific requirements.
Does WeGuide integrate with REDCap and other clinical systems?
Yes. WeGuide's integration engine connects with REDCap, EDC systems, and other eClinical platforms to ensure seamless data flow between your eConsent module and your broader clinical trial technology stack. Contact our team for specific integration requirements.
How do I update consent forms for protocol changes or sub-studies?
WeGuide supports dynamic consent updates. When your protocol evolves or a sub-study is launched, you can add new consent steps or modify existing ones directly in the platform. Affected participants are automatically notified to re-consent, keeping your study compliant without manual tracking.
Can WeGuide collect consent in multiple languages?
Yes. WeGuide supports multi-language consent collection, allowing you to engage participants from diverse linguistic backgrounds. Consent forms can be deployed in multiple languages simultaneously, ensuring accessibility and regulatory compliance across global, multi-site clinical trials.
How is participant data stored and protected?
All participant data is stored on medically certified infrastructure compliant with TGA Class 1, ISO 27001, HIPAA, and 21 CFR Part 11 standards. WeGuide uses advanced encryption, strict access controls, and a complete audit trail to safeguard sensitive clinical research data. Our platform also supports GDPR requirements for studies involving European participants.
How does WeGuide ensure participants understand the consent process?
WeGuide includes built-in comprehension assessments — interactive quizzes and knowledge checks — embedded within the eConsent form. Participants must demonstrate understanding of key study information before they can provide their electronic signature. This reduces the risk of protocol deviations caused by incomplete participant comprehension.
Can I track consent status across my study?
Yes. WeGuide provides a real-time consent tracking dashboard. Monitor which participants have completed consent, which have pending re-consents, and which need reminders — all from a single view. The complete audit trail is available for sponsor review and regulatory audits at any time.
Does WeGuide support remote and decentralized consent?
Yes. WeGuide's eConsent module supports in-person, remote, and hybrid consent workflows. Participants can review and sign consent forms on any device from any location, making it ideal for decentralized clinical trials (DCTs) and studies with geographically dispersed participant populations.
How quickly can I implement eConsent in my trial?
WeGuide's eConsent solution is designed for rapid deployment. Most studies can go live within days, not weeks. Our team provides hands-on support from consent form design through launch, ensuring your eConsent workflow is configured, tested, and compliant before your first participant enrolls.
Does eConsent support signatures from Legally Authorized Representatives?
Yes. WeGuide supports consent workflows that require multiple signatories, including parents, guardians, and Legally Authorized Representatives (LARs). Each signatory can review and sign the consent form independently, whether on-site or remotely.
How do participants complete the eConsent form?
Participants access the eConsent form as a task within their WeGuide program. They can start at their convenience, review interactive materials (text, video, quizzes), and provide their electronic signature at the end. Once the eConsent is completed, additional study tasks — such as baseline questionnaires or screening assessments — are automatically unlocked, creating a seamless participant journey.
Never miss an update
Get all the latest news, blog posts and product updates from WeGuide, delivered directly to your inbox. We'll rarely send more than one email a month.
By clicking Organise a demo, you're confirming that we can contact you to set up a demonstration.
Certified and Compliant by Leading Standards










%201.avif)





