Using the Parkinson's Disease Questionnaire-39 (PDQ-39)
The PDQ-39 is a self-report survey assessing quality of life in people with Parkinson's disease. It covers eight areas of daily living to help doctors understand the condition's impact.


Parkinson's Disease Questionnaire-39 (PDQ-39)
The Parkinson's Disease Questionnaire-39 (PDQ-39) is a specific self-report tool for assessing health-related quality of life. Covering eight dimensions like mobility and emotional well-being, it helps clinicians understand daily challenges. This instrument is essential for tracking progression and guiding care.
Category
Disease
Source
What is Parkinson's Disease Questionnaire-39 (PDQ-39)

The Parkinson's Disease Questionnaire-39 (PDQ-39) stands as the leading tool for checking health-related quality of life in people living with Parkinson's. This self-report survey features 39 questions that look at how the condition impacts a person over the last month. It captures the patient's own view of their overall health status. It covers eight specific areas like mobility, activities of daily living, emotional well-being, and stigma. Patients answer on a five-point scale ranging from never to always, allowing clinicians to spot specific physical or social issues clearly. Taking about 10 to 15 minutes to finish, the PDQ-39 is easy to use in busy clinics or research trials. Doctors use the scores to see if treatments are working well. It is a practical way to measure the full impact of the disease beyond just motor symptoms and guide better care plans.

The PDQ-39 can be scored using two main approaches: calculating specific dimension scores and a single summary index. 1. Dimension Scores: Patients rate items from 0 to 4. For domains like Mobility or Stigma, you sum the points, divide by the maximum possible score, and multiply by 100. This creates a 0 to 100 scale where lower numbers mean better quality of life. 2. Summary Index: This method averages the eight dimension scores into one total figure. It is handy for tracking overall health changes in the clinic without getting lost in the details, giving a quick snapshot of how a patient is travelling.

Advantages
Measures healthcare intervention results systematically.
Strengthens evidence through systematic measurement approaches.
Develops personalised strategies based on assessment data.
Promotes active participation in healthcare assessment.
Related Instruments
Screens for neuropathic pain components.
Predicts long-term disability risk in musculoskeletal pain.
Assesses symptoms and function in lumbar spinal stenosis.
Collect PDQ-39 data with WeGuide, the all in one patient engagement platform
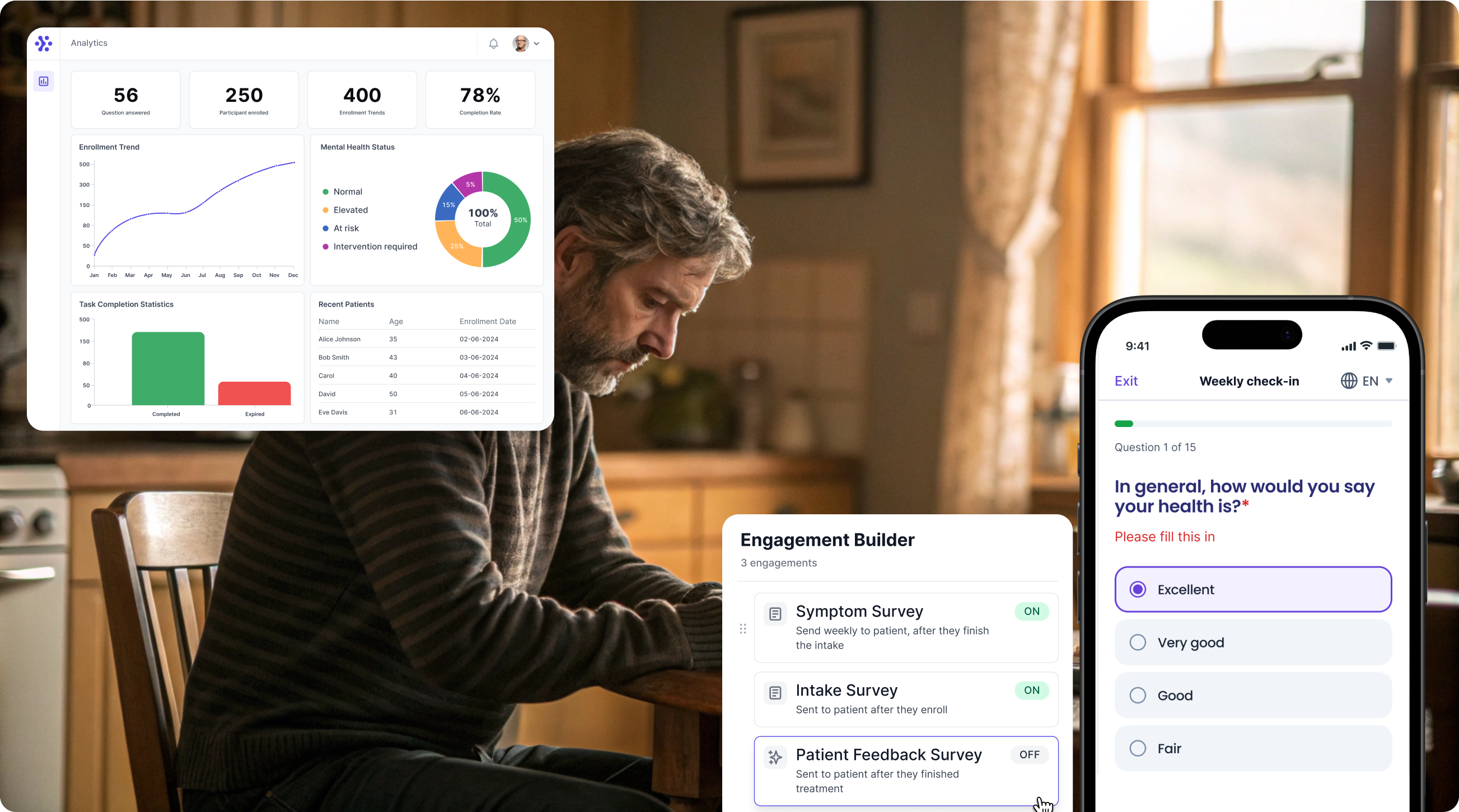

Support Your patient in no time - all under your own brand
Our platform combines the knowledge of 100+ digital health solutions built, letting you easily build your patient engagement app for research or clinical use. Collect PROMs, support patients on waiting lists, or gather vital signs data. This is how:
Learn MoreProgram Builder
.png)
Whitelabelling
.svg)
.svg)
Turn on components, dependent on your usecase
.svg)
Support The Full Journey
.png)
.png)
%20(1).png)
.png)
It’s your turn
Every WeGuide project, saves researchers and clinicians - 9 months and $450,000 - creating impactful digital health solutions.




