Multiple Sclerosis Impact Scale (MSIS-29) Assessment
The MSIS-29 measures the physical and mental impact of multiple sclerosis. This patient survey helps doctors track changes in daily life to support better treatment decisions.


Multiple Sclerosis Impact Scale (MSIS-29)
The Multiple Sclerosis Impact Scale (MSIS-29) is a patient-reported questionnaire assessing the physical and psychological impact of MS. With 29 items, it helps clinicians track disease progression and quality of life. This tool is essential for understanding a patient's daily challenges and adjusting care plans.
Category
Disease
Source
What is Multiple Sclerosis Impact Scale (MSIS-29)

The Multiple Sclerosis Impact Scale (MSIS-29) is a trusted tool for measuring the physical and mental effects of MS. It tracks patient health accurately. This patient-reported outcome measure features 29 specific questions. It focuses on the past two weeks and asks patients to rate their own experiences directly without fuss. The scale splits into two main parts where 20 items cover physical limits and 9 items look at psychological well-being. Patients answer using a simple one to five scale ranging from not at all to extremely, making it easy to spot changes in their condition. Clinicians value this tool because it is quick to finish and scores are simple to calculate. It works well in clinical trials or routine check-ups to monitor disease progression. By capturing the patient's view, it supports better treatment choices and helps improve care plans for the long run.

The MSIS-29 can be scored using two main approaches: raw summing and percentage transformation. 1. Raw Summing: You simply add up the responses for the 20 physical items and 9 psychological items separately. Each question gets rated from 1 for not at all to 5 for extremely, giving you two distinct totals to track health changes. 2. Transformed Score: To make things easier to compare, clinicians often convert these totals to a 0 to 100 scale. You subtract the lowest possible score from the actual score, divide by the range, and multiply by 100. Higher numbers mean the MS is having a bigger impact on daily life.

Advantages
Measures healthcare intervention results systematically.
Provides consistent measurements for clinical research.
Promotes active participation in healthcare assessment.
Strengthens evidence through systematic measurement approaches.
Related Instruments
Screens for neuropathic pain components.
Predicts long-term disability risk in musculoskeletal pain.
Assesses symptoms and function in lumbar spinal stenosis.
Collect MSIS-29 data with WeGuide, the all in one patient engagement platform
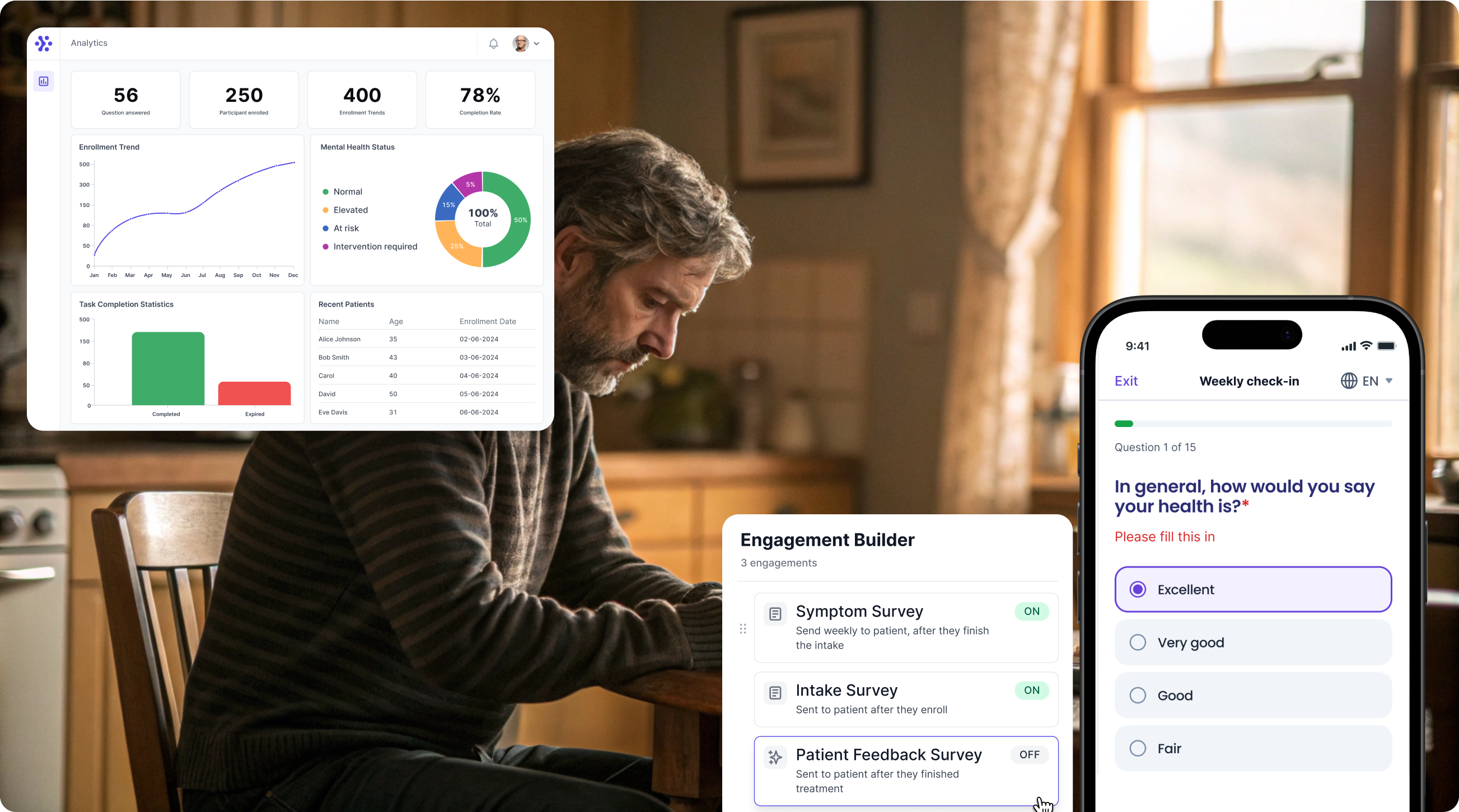

Support Your patient in no time - all under your own brand
Our platform combines the knowledge of 100+ digital health solutions built, letting you easily build your patient engagement app for research or clinical use. Collect PROMs, support patients on waiting lists, or gather vital signs data. This is how:
Learn MoreProgram Builder
.png)
Whitelabelling
.svg)
.svg)
Turn on components, dependent on your usecase
.svg)
Support The Full Journey
.png)
.png)
%20(1).png)
.png)
It’s your turn
Every WeGuide project, saves researchers and clinicians - 9 months and $450,000 - creating impactful digital health solutions.




