Modified Rankin Scale (mRS): Measuring Stroke Disability
Clinicians use the Modified Rankin Scale to assess disability after a stroke. It measures a patient's independence in daily tasks, helping care teams track recovery progress over time.


Modified Rankin Scale (mRS)
The Modified Rankin Scale (mRS) measures the degree of disability or dependence in daily activities for stroke survivors. Ranging from 0 to 6, it helps doctors assess recovery outcomes. It's a key tool for tracking how much assistance a patient needs after a neurological event.
Category
Disease
Source
What is Modified Rankin Scale (mRS)

The Modified Rankin Scale (mRS) is a trusted tool for measuring disability levels after a stroke. It helps doctors see how well patients recover daily. This scale looks at global disability and focuses on how much help a person needs. It serves as the standard way to track neurological function and independence outcomes over time. The mRS uses a range from 0 to 6. Zero means no symptoms at all, while five indicates severe disability requiring constant nursing care. A score of six represents death. This setup helps clinicians spot changes in a patient's ability to handle daily tasks. Clinicians or researchers can run this test quickly through an interview. It fits well in hospital settings or clinical trials because it is easy to use and reliable. By understanding these scores, healthcare teams can plan better rehab goals and support patients as they get back to their lives.

The Modified Rankin Scale can be scored using one main approach: a global disability grade. Clinicians assign a single number from 0 to 6 based on a structured assessment of how well a patient handles daily tasks. A score of 0 means no symptoms at all, while numbers climb to 5 for severe bedridden disability requiring constant nursing care. A 6 indicates death. For research, experts often group scores 0 to 2 as a functional independence outcome to check if treatments work. This helps teams spot important changes in stroke recovery over time.

Advantages
Measures healthcare intervention results systematically.
Meets international clinical assessment requirements.
Strengthens evidence through systematic measurement approaches.
Streamlines research data gathering process.
Related Instruments
Screens for neuropathic pain components.
Predicts long-term disability risk in musculoskeletal pain.
Assesses symptoms and function in lumbar spinal stenosis.
Collect Modified Rankin Scale data with WeGuide, the all in one patient engagement platform
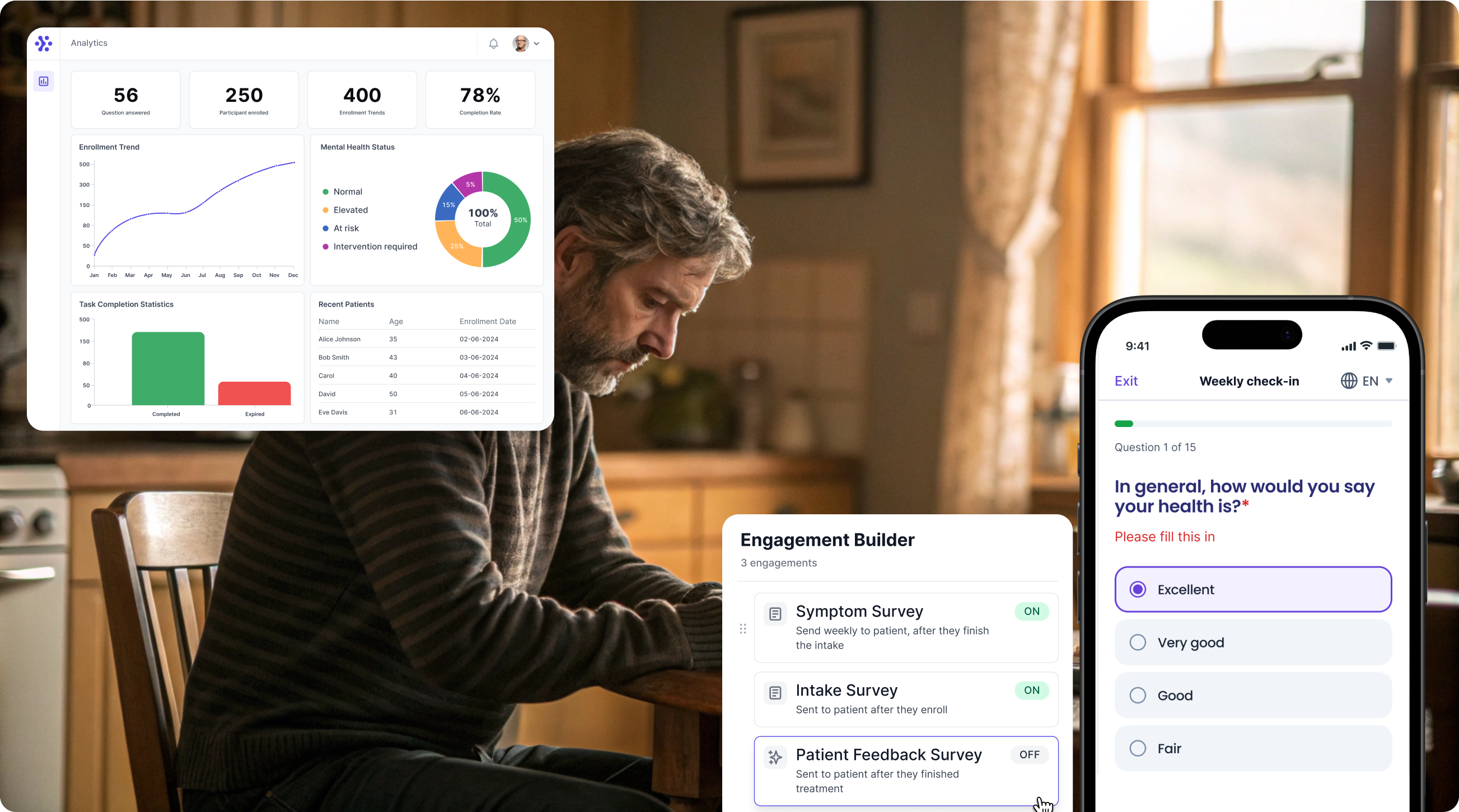

Support Your patient in no time - all under your own brand
Our platform combines the knowledge of 100+ digital health solutions built, letting you easily build your patient engagement app for research or clinical use. Collect PROMs, support patients on waiting lists, or gather vital signs data. This is how:
Learn MoreProgram Builder
.png)
Whitelabelling
.svg)
.svg)
Turn on components, dependent on your usecase
.svg)
Support The Full Journey
.png)
.png)
%20(1).png)
.png)
It’s your turn
Every WeGuide project, saves researchers and clinicians - 9 months and $450,000 - creating impactful digital health solutions.




