Bath Ankylosing Spondylitis Disease Activity Index (BASDAI)
The BASDAI is a self-reported survey that tracks disease activity in people with ankylosing spondylitis. It measures symptoms like fatigue and spinal pain to help doctors manage treatment.


Bath Ankylosing Spondylitis Disease Activity Index (BASDAI)
The Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) is a trusted scale for measuring disease activity in ankylosing spondylitis. It uses six questions on fatigue, pain, and stiffness to generate a score out of 10. This helps clinicians track symptom changes and manage therapy based on patient input.
Category
Disease
Source
What is Bath Ankylosing Spondylitis Disease Activity Index (BASDAI)

The Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) is the standard method for tracking active disease in people with spondyloarthritis. It relies on six specific questions that patients answer themselves. This simple setup helps doctors see how the condition impacts daily life without needing complex lab tests. The survey looks at major symptoms like fatigue and spinal pain plus joint swelling and localised tenderness. It also checks morning stiffness severity and duration. Patients rate their discomfort on a zero to ten scale, where ten indicates very severe symptoms. Finishing the survey takes less than two minutes, making it perfect for busy clinics. A score above four usually suggests active inflammation, helping teams decide if new therapies are needed. It's a reliable way to monitor changes over time and guide better treatment choices for long-term care.

The BASDAI is scored using a specific formula that turns patient answers into a single number tracking disease activity. Patients rate six symptoms, including fatigue and spinal pain, on a 0 to 10 scale. To get the score, find the mean of the two morning stiffness questions and add it to the first four items. Divide this sum by 5 to get a final result between 0 and 10. A score of 4 or more usually points to active disease, which helps your specialist track how well treatments are working over time.

Advantages
Tracks patient condition changes systematically.
Develops personalised strategies based on assessment data.
Meets international clinical assessment requirements.
Streamlines research data gathering process.
Related Instruments
Screens for neuropathic pain components.
Predicts long-term disability risk in musculoskeletal pain.
Assesses symptoms and function in lumbar spinal stenosis.
Collect BASDAI data with WeGuide, the all in one patient engagement platform
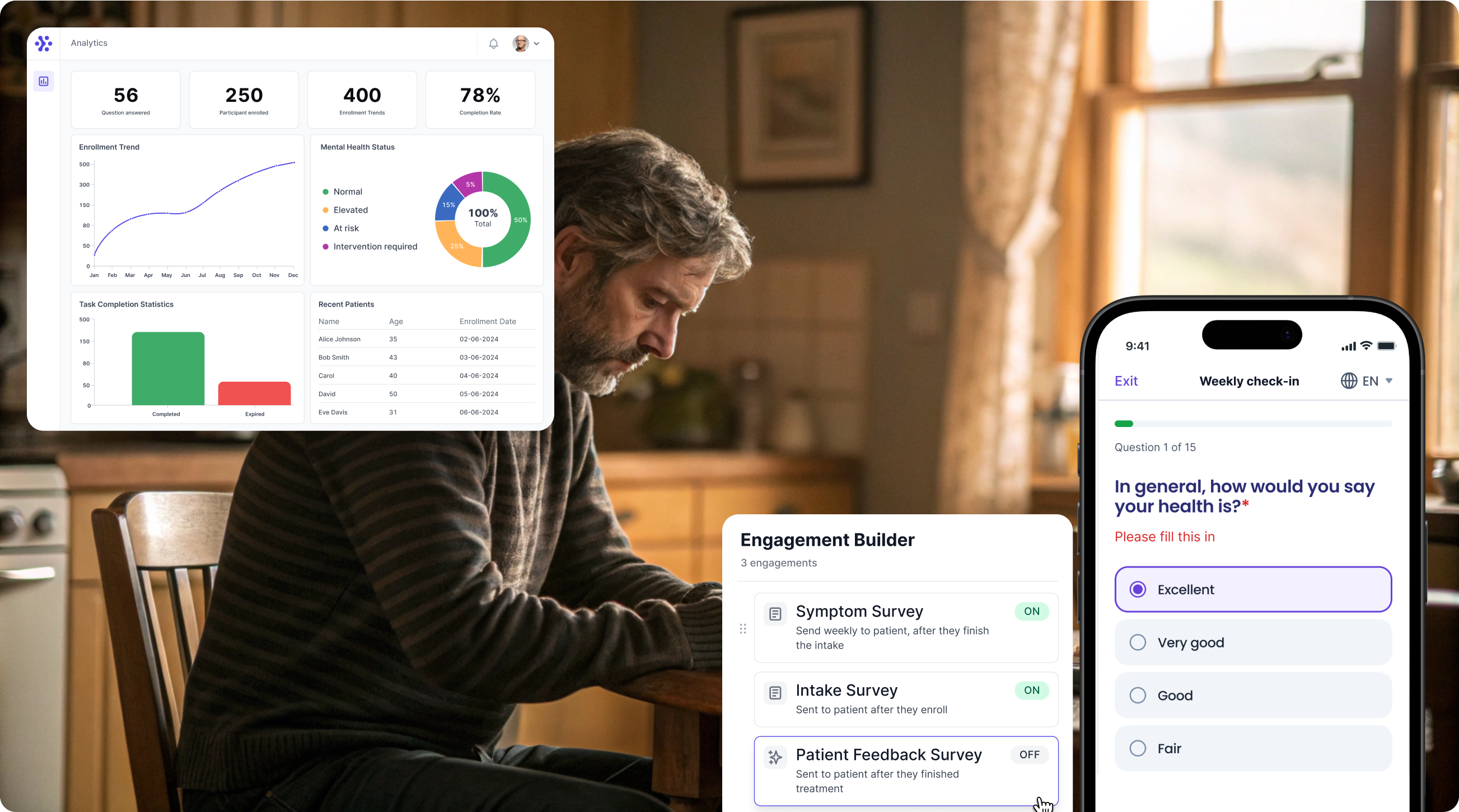

Support Your patient in no time - all under your own brand
Our platform combines the knowledge of 100+ digital health solutions built, letting you easily build your patient engagement app for research or clinical use. Collect PROMs, support patients on waiting lists, or gather vital signs data. This is how:
Learn MoreProgram Builder
.png)
Whitelabelling
.svg)
.svg)
Turn on components, dependent on your usecase
.svg)
Support The Full Journey
.png)
.png)
%20(1).png)
.png)
It’s your turn
Every WeGuide project, saves researchers and clinicians - 9 months and $450,000 - creating impactful digital health solutions.




