Asthma Control Questionnaire (ACQ): Symptom Assessment
The Asthma Control Questionnaire helps doctors and patients measure how well asthma is controlled. It assesses symptoms and medication use to provide a score that guides treatment decisions.


Asthma Control Questionnaire
The Asthma Control Questionnaire (ACQ) assesses how well a patient's asthma is managed. By tracking symptoms and lung function, it helps clinicians adjust treatments to minimise risk. This survey provides a clear score, ensuring respiratory health is monitored accurately in clinical practice.
Category
Disease
Source
What is Asthma Control Questionnaire

The Asthma Control Questionnaire (ACQ) is a trusted tool for checking asthma management. It helps doctors and patients track symptom stability. This validated survey looks at the main criteria for stable asthma. It mixes subjective patient reports with objective lung function data for a full view. Patients answer questions about waking up at night, morning symptoms, activity limits, shortness of breath, and wheezing. They also record rescue bronchodilator use. All items use a seven point scale where zero means well controlled and six means poor control. Clinicians use the full ACQ with seven items or shorter versions like the ACQ-5 that skip the lung function test. It is useful for clinical trials and routine visits because it spots health changes quickly. The score helps teams decide if they need to change treatment plans to keep lungs healthy.

The Asthma Control Questionnaire can be scored using a straightforward averaging approach. Patients answer questions on a 7 point scale ranging from 0 (no impairment) to 6 (maximum impairment). To get the final score, you just add up the response values and divide by the total number of questions answered. This gives a mean score between 0 and 6. Generally, a score below 0.75 suggests asthma is well controlled, while anything over 1.5 indicates it's not well controlled. Scores sitting in between fall into a grey area requiring clinical judgement.

Advantages
Tracks patient condition changes systematically.
Develops personalised strategies based on assessment data.
Provides consistent measurements for clinical research.
Strengthens evidence through systematic measurement approaches.
Related Instruments
Screens for neuropathic pain components.
Predicts long-term disability risk in musculoskeletal pain.
Assesses symptoms and function in lumbar spinal stenosis.
Collect Asthma Control Questionnaire data with WeGuide, the all in one patient engagement platform
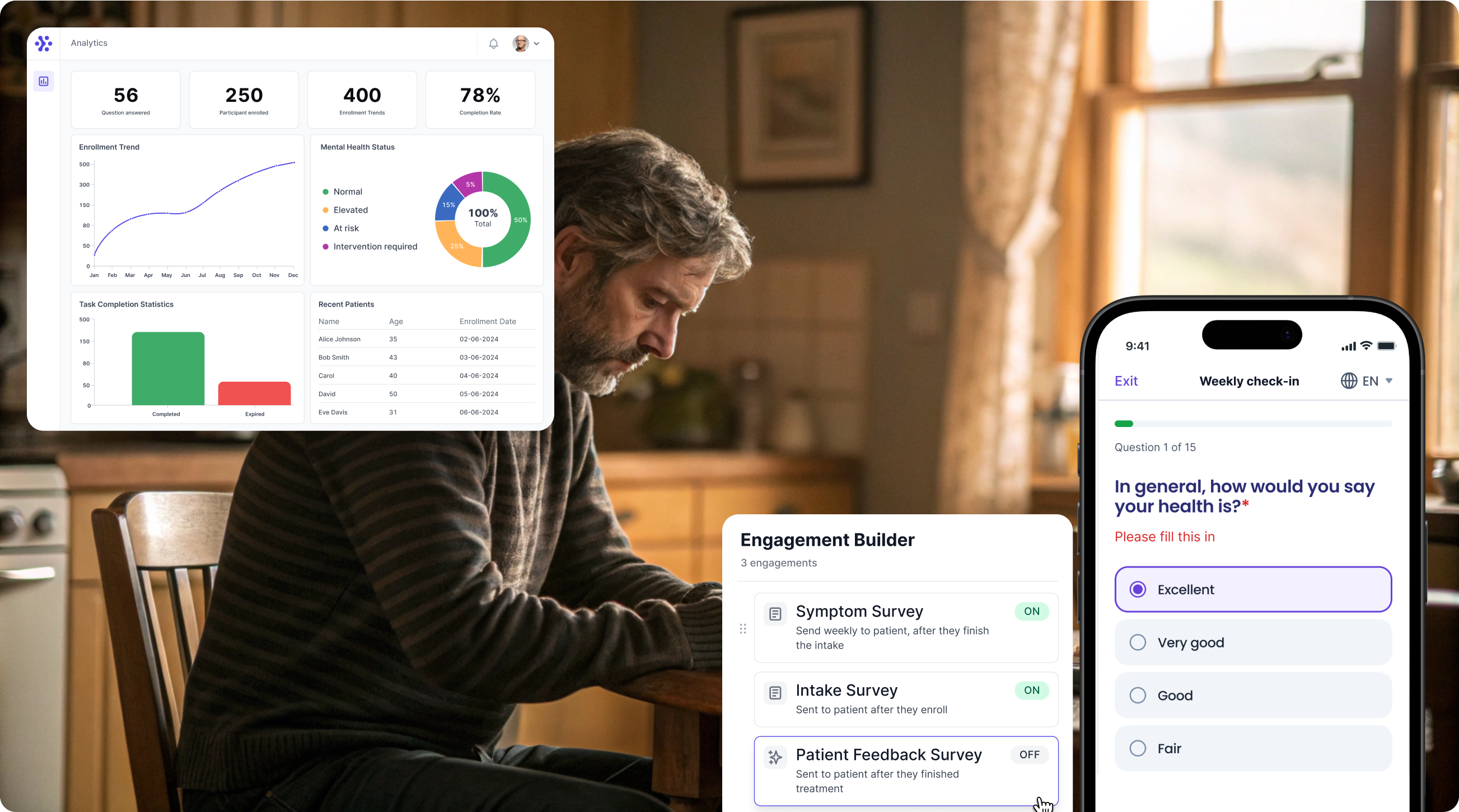

Support Your patient in no time - all under your own brand
Our platform combines the knowledge of 100+ digital health solutions built, letting you easily build your patient engagement app for research or clinical use. Collect PROMs, support patients on waiting lists, or gather vital signs data. This is how:
Learn MoreProgram Builder
.png)
Whitelabelling
.svg)
.svg)
Turn on components, dependent on your usecase
.svg)
Support The Full Journey
.png)
.png)
%20(1).png)
.png)
It’s your turn
Every WeGuide project, saves researchers and clinicians - 9 months and $450,000 - creating impactful digital health solutions.




