WeGuide alternatives: compare 12 clinical trial platforms (2026)
WeGuide is the patient-first clinical research platform that replaces three to five stitched tools with one, deploys in 6 weeks, and holds 94% average participant adherence across 200,000+ participants. Whether you're outgrowing REDCap, frustrated with OpenClinica's setup, or evaluating enterprise platforms like Medidata, Veeva, Oracle, IQVIA, or Clario, this hub walks through how WeGuide compares across every dimension that matters.
No pressure, no hard sell. A 30-minute walkthrough with a research colleague who's built platforms like yours. Cancel anytime, no commitment.
%20(1).png)
Used by leading research and medical institutes.








%201.avif)










Platform choice has a compounding effect on a study. The right fit reduces implementation weeks, lifts participant adherence, and removes the procurement friction of stitching five tools together.
Most evaluations land on one of three questions:
"We're outgrowing REDCap or OpenClinica." You need eCOA, eConsent, wearables, and a participant mobile app that open-source tools weren't built for.
"We're priced out of Medidata, Veeva, Oracle, or Medable." Enterprise platforms bill at six and seven figures with 3-to-6-month implementations. Mid-market research budgets don't absorb that.
"We need a technology platform, not a CRO services contract." IQVIA, Science 37, Thread, and ObvioHealth bundle services with the platform. For teams that own study operations or already work with a CRO, a pure technology platform is a cleaner fit.
WeGuide sits in a specific place in this market: a patient-first clinical research platform with whitelabel mobile apps, native wearable integration, and a 200+ validated instrument library, priced for mid-market sponsors, academic research, CROs, and health organisations. It isn't a replacement for every platform below, and we'll say so wherever it's true.
%20(1).png)
When the use case matches, here's what changes for research teams who move to WeGuide.
.png)
94% average adherence, proven at scale
.svg)
Deploy in 6 weeks, not 6 months
.png)
One platform, not five stitched tools
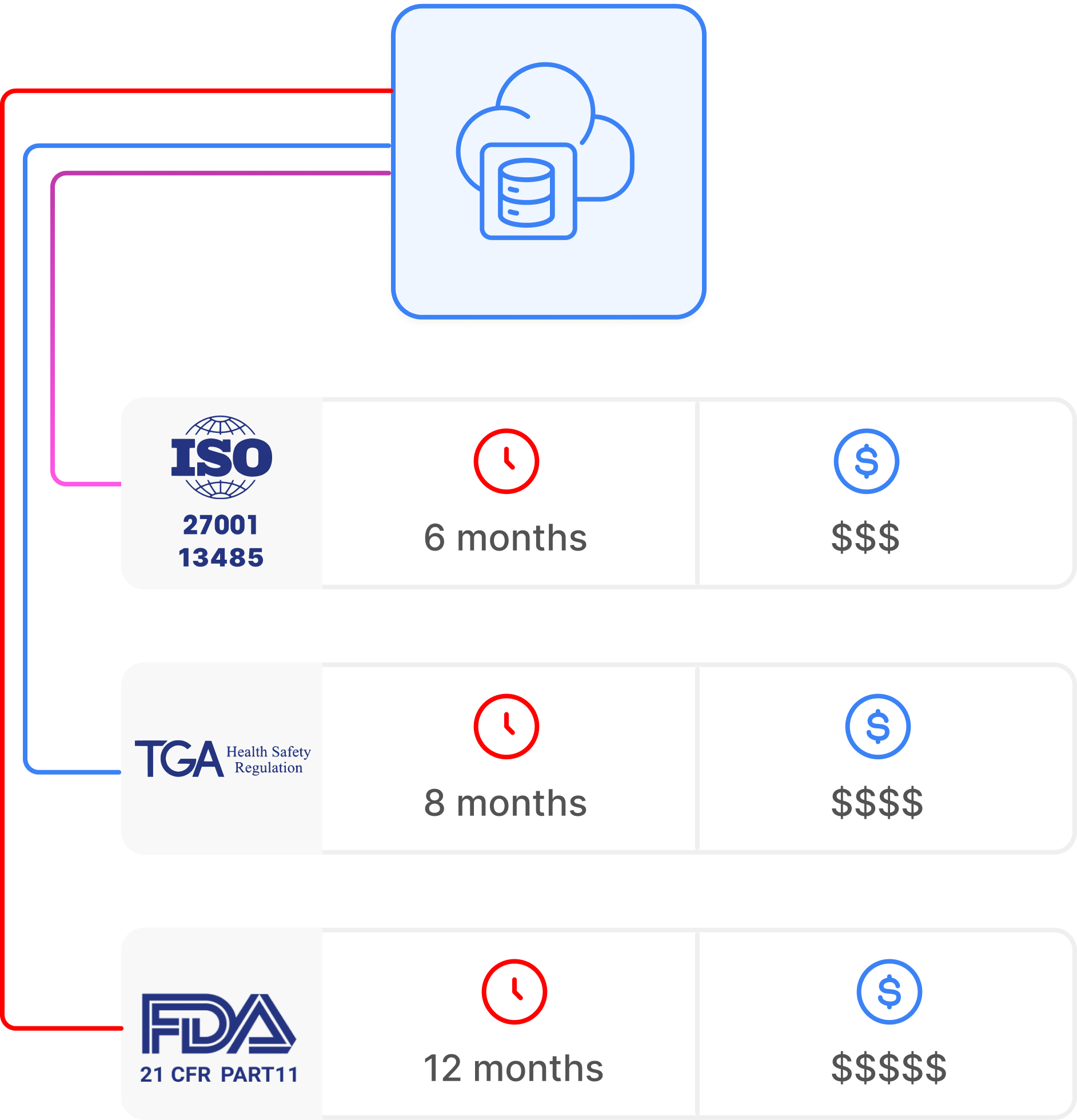
Compliance built in from day one
.png)

.png)

.png)

BRACE Trial (Murdoch Children's Research Institute)
A global randomised controlled trial studying whether the BCG vaccine could reduce COVID-19 symptom severity. Launched across five countries with 6,000+ participants in six weeks during the pandemic. Adherence exceeded 90%. The level of clinical trial delivery enterprise DCT vendors charge pharma budgets to match.
GenV (Generation Victoria)
A population health registry tracking more than 100,000 Victorian families from birth. Longitudinal data collection, multi-year participant relationships, and a family-branded whitelabel app. Registry work that most DCT-focused competitors don't offer natively.
FSHD Global Registry
A rare disease patient registry for facioscapulohumeral muscular dystrophy, running globally with longitudinal participant engagement. Another registry use case outside DCT-only competitors' scope.
Which clinical trial platform is the best fit?
Platform choice is study specific. Here's the honest guidance we give teams on discovery calls.
Best for academic research and hospital-led studies → WeGuide (whitelabel apps, managed cloud, no institutional IT burden) or REDCap (if no engagement or wearables needed)
Best for enterprise pharma Phase II-IV with hundreds of sites → Medidata Rave or Veeva Vault EDC
Best for mid-market CROs and sponsors → WeGuide (accessible pricing, 6-week deployment)
Best for decentralised clinical trials at top-50 pharma budgets → Medable or Thread Research
Best for nutraceutical and consumer health DCTs with bundled services → ObvioHealth
Best for trials needing mobile nurses or virtual site visits → Science 37 (or successor) for services; WeGuide for technology
Best for specialised cardiac safety, imaging, or respiratory endpoints → Clario
Best for site-side eBinders and eRegulatory → Florence Healthcare (complementary to WeGuide)
Best for pharmacovigilance and Argus-connected submissions → Oracle Health Sciences
Best for CRO-bundled full-service trial execution → IQVIA
Best for outcome measure design consulting → Thread Research (via Modus Outcomes)
Best for patient registries, population health, employee wellbeing, and citizen science → WeGuide (first-class use cases)
Frequently asked questions
Straight answers on how WeGuide compares across the major clinical trial platforms.
How does WeGuide compare to REDCap?
REDCap is free and excellent for academic survey research with 7,000+ institutions. WeGuide adds a whitelabel participant mobile app, native wearable integration (Garmin, Apple Health, Google Fit), built-in eConsent with version control, TGA Class I certification, and multilingual translation management. For studies that need engagement, eCOA, eConsent, or wearables, WeGuide extends beyond REDCap's academic survey scope. For pure data collection on a zero-dollar budget with institutional IT support, REDCap is still a strong choice and we'll say so. See the full REDCap comparison →
How does WeGuide compare to OpenClinica?
OpenClinica is an EDC first open source platform with strong eCRF design and edit check capability. WeGuide is patient-first, with EDC available via Integration Engine rather than natively. The two are complementary for studies that combine deep EDC requirements with participant engagement. For pure site-based EDC with eCRFs, OpenClinica is hard to beat. For ePRO, eConsent, wearables, patient registries, or employee wellbeing, WeGuide covers the ground OpenClinica doesn't.
Which platforms support native wearable data collection?
WeGuide integrates natively with Garmin Health, Apple Health, and Google Fit through first party SDK work. Most competitors (Medable, Medidata, Veeva, Oracle, Thread, ObvioHealth) support wearables via partner integrations or connected device layers. Clario provisions validated devices rather than supporting BYOD wearables. For BYOD wearable data collection in a clinical research context, WeGuide's native SDK approach is unusual in the market.
Is WeGuide FDA 21 CFR Part 11 and TGA compliant?
Yes. WeGuide is TGA certified as Class I medical device software, ISO 27001 certified, FDA 21 CFR Part 11 ready, HIPAA and GDPR compliant, and CE marked. Australian data residency is a first-class offering for APAC sponsors and academic research.
How quickly can I deploy a study with WeGuide?
Typical deployment is six weeks. The BRACE Trial launched across five countries with 6,000+ participants in six weeks during the pandemic. Enterprise platforms (Medidata, Veeva, Oracle, Medable) typically measure implementation in three to six months with a services engagement. The no code Form Builder lets research coordinators configure studies directly rather than handing specs to a services team.
Can WeGuide handle multilingual studies?
Yes. INHERIT ran in five languages with dynamic translation management and culturally adapted content during COVID-19. Multilingual translation management is built into the platform rather than requiring per-language survey configuration. GenV runs across Victoria's multilingual families on the same infrastructure.
Does WeGuide integrate with existing EDCs?
Yes. Integration Engine connects to Medidata Rave, Veeva Vault, REDCap, Viedoc, Clinion, Oracle InForm, and custom EDCs. For sponsors who already have an EDC preference or want flexibility, WeGuide runs alongside rather than replacing. Many teams pair WeGuide (participant-facing) with their existing EDC (data management).
How does pricing compare across clinical trial platforms?
Enterprise platforms (Medidata, Veeva, Oracle, Medable, Clario, IQVIA) bill at six and seven figures with multi year contracts, often bundled with services. Services-led platforms (Science 37, ObvioHealth, Thread) bundle virtual CRO or consulting scope into the quote. WeGuide is technology only project-based pricing with every module included in the core licence, priced accessibly for mid-market sponsors, academic research, and CROs. REDCap and OpenClinica community edition are free but require institutional IT to run.
Never miss an update
Get all the latest news, blog posts and product updates from WeGuide, delivered directly to your inbox. We'll rarely send more than one email a month.
By clicking Organise a demo, you're confirming that we can contact you to set up a demonstration.
Certified and Compliant by Leading Standards










%201.avif)





